National COVID-19 Case Surveillance
To protect Americans from serious infectious diseases and other health threats, public health authorities conduct national case surveillance to monitor more than 120 diseases and conditions. For these conditions, public health collects information on individuals with the infection in a population, which is known as case surveillance. The goal of case surveillance is to provide information necessary to control outbreaks and inform public health action. Case surveillance is especially important for new diseases, such as COVID-19, in order to understand the similarities and differences between cases, including:
- demographic, clinical, and epidemiologic characteristics,
- exposure and contact history, and
- course of clinical illness and care received.
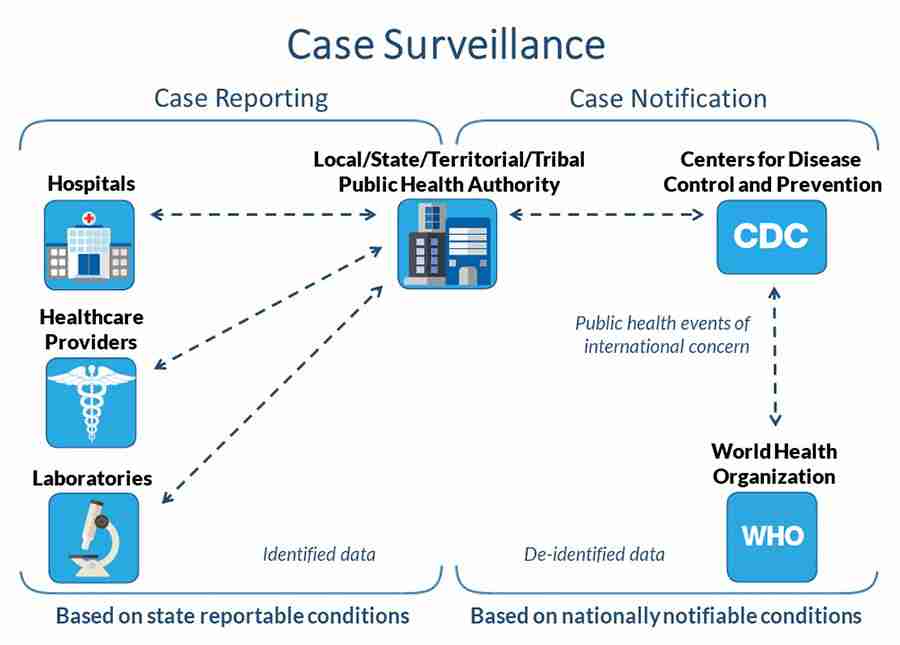
During the COVID-19 response, state and jurisdictional health departments voluntarily report to CDC using the National Notifiable Diseases Surveillance System. Data about COVID-19 cases are sent to CDC without personal identifiers, such as names or home addresses. A national standardized case definition is used to define confirmed and probable cases and deaths.
Unlike data collected for clinical trials and research studies in which scientists comprehensively measure and follow the health status of a patient, national case surveillance data focuses on capturing demographic and risk factor information about people with COVID-19.
The process for reporting, collecting, and analyzing disease data may be called a data supply chain. Under state disease reporting laws hospitals, healthcare providers, and laboratories must report confirmed or probable COVID-19 cases and deaths to state or local health departments. These laws allow health departments to quickly identify outbreaks and control the spread of disease. The figure illustrates how data is transferred for case reporting (from hospitals, healthcare provider, and laboratories to local, regional, state, or territorial public health) and how data moves for case notification (from state or territorial public health to CDC). These two steps of information flow make up national case surveillance. While case reporting is mandatory under state reportable disease laws, case notification from state and local health departments to CDC includes deidentified data and is voluntary.
Using the National Notifiable Diseases Surveillance System, health departments voluntarily send CDC data that does not include personal, identifiable information about COVID-19 cases and deaths. Because COVID-19 has been designated as a public health emergency of international concern, CDC reports national case surveillance data to the World Health Organization under International Health Regulations (2005). CDC also publishes deidentified COVID-19 national case surveillance data with additional privacy protections for public use at data.cdc.gov.
National case surveillance does not provide real-time data, which can be provided by other data sources such as syndromic surveillance, a process monitoring the types of symptoms people have when they go to emergency departments. However, case surveillance provides information on the characteristics of a disease within a population, usually with laboratory-confirmation of cases and following a standard case definitionpdf iconexternal icon. CDC uses national case surveillance to:
- Track the spread of COVID-19 around the country to identify areas of concern and inform state decision makers.
- Help state and local public health departments better control COVID-19 by evaluating trends in demographics, exposures, and outcomes among cases in order to identify those groups most at risk, such as health care workers, racial/ethnic minorities, older adults, and people with underlying health conditions.
- Analyze exposure information and health outcomes among COVID-19 patients to develop guidance for the public, at-risk groups, and healthcare providers.
The COVID-19 pandemic has put unprecedented strain on the public health data supply chain. In many states, the large number of COVID-19 cases has severely strained the ability of hospitals, healthcare providers, and laboratories to report cases with complete demographic information, such as race and ethnicity. The unprecedented volume of cases has also limited the ability of state and local health departments to conduct thorough case investigations and collect all requested case data. As a result, many COVID-19 case reports submitted to CDC do not have complete information on patient demographics, clinical outcomes, exposures and factors that may put people at higher risk for severe disease.
National case surveillance data are constantly changing. For instance, as new information is gathered about previously reported cases, health departments provide updated data to CDC. As more complete information becomes available, analytic updates might capture changes in surveillance data and trends during a previously reported time window.
A limitation of case reporting is that people who are infected with the virus that causes COVID-19 may have mild or no symptoms. These people may not seek testing or healthcare and are therefore less likely to be reported. Similarly, cases where individuals have had severe outcomes, such as hospitalizations, ICU admissions, and deaths, are more likely to be reported than outcomes that are less severe.
CDC continues to work with state, local, and territorial health departments to accelerate reporting of national case surveillance data, improve data quality, and gather complete information about all COVID-19 cases.
CDC is working with healthcare providers, electronic health record developers, laboratories, and state and local health departments to modernize disease surveillance by automating the generation and transmission of case reports from the electronic health record to public health agencies for review and action for the COVID-19 response.
For example, expanded use of electronic case reporting, which make the submission of information from healthcare providers to public health departments seamless and automated, will reduce the burden of manually reporting COVID-19 cases, increase timeliness of reporting, and improve data completeness by pulling data directly from the medical record.
Additional resources
- CDC COVID Data Tracker
- CDC’s National Notifiable Diseases Surveillance System
- Electronic Case Reporting for COVID-19
- CSTE Case definition for COVID-19pdf iconexternal icon
- International Health Regulations (2005)
- Public Health Surveillance in the United States: Evolution and Challenges, June 2012
- Modernizing Centers for Disease Control and Prevention Informatics Using Surveillance Data Platform Shared Servicesexternal icon March—April, 2018
- CDC’s Vision for Public Health Surveillance in the 21st Centurypdf icon, July 2012
- Centers for Disease Control and Prevention (CDC). Introduction to Public Health. In: Public Health 101 Series. Atlanta, GA: U.S. Department of Health and Human Services, CDC; 2014.
CDC COVID-19 Surveillance
Public health surveillance is the ongoing, systematic collection, analysis, and interpretation of health-related data essential to planning, implementation, and evaluation of public health practice.
For surveillance of COVID-19 and the virus that causes it, SARS-CoV-2, CDC is using multiple surveillance systems in collaboration with state, local, territorial, academic, and commercial partners to monitor COVID-19 in the United States. COVID-19 surveillance draws from a combination of data sources using existing influenza and viral respiratory disease surveillance, syndromic surveillance, case reporting, lab reporting, the healthcare safety system, ongoing research platforms, and other new surveillance systems designed to answer specific questions. The data from these systems, combined, create an updated picture of COVID-19’s spread and its effects in the United States and are used to inform the U.S. national public health response to COVID-19.
- To monitor spread of COVID-19 in the United States
- To understand disease severity and the spectrum of illness due to COVID-19
- To understand risk factors for severe disease and transmission of COVID-19
- To monitor for changes in the virus that causes COVID-19
- To estimate disease burden due to COVID-19
- To produce data for forecasting COVID-19’s spread and impact
- To understand how COVID-19 impacts the capacity of the U.S. healthcare system (for example, availability and shortages of key resources)
COVID-19 data can be used to help public health professionals, policy makers, and health care providers monitor the spread of COVID-19 in the United States and support better understanding of illness, disease severity, effectiveness of community interventions, and social disruptions associated with COVID-19 in the U.S.. These data help inform U.S. national, state, local, tribal, and territorial public health responses to COVID-19.
Detailed and accurate data will allow us to better understand and track the size and scope of the outbreak and strengthen prevention and response efforts.
CDC provides this information on the Cases, Data, & Surveillance webpage:
-
Cases in the U.S.
- Cases reported in the U.S. so far (and new cases reported in the past day)
- Deaths reported in the U.S. so far (and new deaths reported in the past day)
-
Cases & Deaths by County
- Reported deaths by county in the U.S. so far (and new deaths reported in the past day)
-
COVIDView: A Weekly Surveillance Summary of COVID-19 Activity
- Key updates for the week (trends in laboratory testing, outpatient and emergency department visits, hospitalizations, and deaths)
- U.S. Virologic Surveillance (national percentage of respiratory specimens testing positive for SARS-CoV-2 at public health, clinical, and commercial laboratories)
- Outpatient and emergency department visits
- Cumulative COVID-19-associated hospitalization rates and patient characteristics (age, race/ethnicity, and underlying medical conditions)
- National percentage of deaths attributed to pneumonia, influenza, or COVID-19
-
CDC COVID Data Tracker
- U.S. COVID-19 cases and deaths by state/territory (counts, rates, and deaths by age group and sex)
- U.S. laboratory testing for COVID-19
- U.S. COVID-19 cases and deaths by county
- Social impact COVID-19 events by state (for example, school closures, localized outbreaks, and state of emergency declarations)
- Human mobility and transmission by state, county, and county classification (workplace, retail and recreation, transit station, and at home mobility tracking)
-
Hospitalization Surveillance Network (COVID-NET)
- National hospitalization rates for COVID-19
- Characteristics of people hospitalized with COVID-19 in the U.S.
-
COVID-19 Serology Surveillance
- Information on large-scale geographic, community-level, and special populations seroprevalence surveys (Results from these surveys will be posted as they become available.)
-
COVID-19 Data from the National Center for Health Statistics (NCHS)
- National Vital Statistic System’s provisional death counts
- Data on mental health and access to healthcare from the NCHS partnership with the U.S. Census Bureau on the Household Pulse Survey (includes indicators of anxiety and depression based on reported frequency of symptoms during the last 7 days)
-
Testing Data in the U.S.
- Number of tests (viral and antibody) conducted and positive tests reported in the U.S.
-
Patient Impact and Hospital Capacity Pathway
- Estimated percentage of inpatient beds occupied by all patients, by state
- Estimated percentage of inpatient beds occupied by COVID-19 patients, by state
- Estimated percentage of ICU beds occupied by all patients, by state
Understanding the Data
A COVID-19 case includes confirmed and probable cases and deaths. This change was made to reflect an interim COVID-19 positionpdf iconexternal icon statement issued by the Council of State and Territorial Epidemiologists on April 5, 2020. The position statement included a case definition and made COVID-19 a nationally notifiable disease.
A probable case or death is defined as:
- A person meeting clinical criteria AND epidemiologic evidence with no confirmatory laboratory testing performed for COVID-19;
- A person meeting presumptive laboratory evidence AND either clinical criteria OR epidemiologic evidence;
- A person meeting vital records criteria with no confirmatory laboratory testing performed for COVID-19.
The rise in the number of COVID-19 cases reflects the rapid community spread of COVID-19 in many U.S. states, territories and communities. Community spread means people have been infected with the virus in an area, including some who are not sure how or where they became infected. Also, the number of cases of COVID-19 being reported in the United States is rising due to increased laboratory testing and reporting across the country.
The COVID-19 death count shown on the Cases in the U.S. web page includes deaths reported daily by state, local, and territorial health departments. This count reflects the most up-to-date information received by CDC based on preliminary reporting from health departments.
In contrast, provisional COVID-19 death counts from the National Center for Health Statistics (NCHS) are updated Monday through Friday with information collected from death certificates. These data represent the most accurate death counts. However, because it can take several weeks for death certificates to be submitted and processed, there is on average a delay of 1–2 weeks before they are reported. Therefore, the provisional death counts may not include all deaths that occurred during a given time period, especially for more recent periods. Death counts from earlier weeks are continually revised and may increase or decrease as new and updated death certificate data are received. Provisional COVID-19 death counts may therefore differ from those on other published sources, such as media reports or the Cases in the U.S. web page.
The mortality rate is the percentage of people who died due to COVID-19 out of the total number of people with COVID-19 reported. Since this is an ongoing outbreak, the percentage can change daily. There are several reasons for this, such as there may be delays in reporting of additional confirmed cases of COVID-19 and not all COVID-19 cases will be detected.
CDC’s overall case numbers are validated through a confirmation process with jurisdictions. The process used for finding and confirming cases displayed by different places may differ.
Case numbers reported on other websites may differ from what is posted on CDC’s website because CDC’s overall case numbers are validated through a confirmation process with each jurisdiction. Differences between reporting jurisdictions and CDC’s website may occur due to the timing of reporting and website updates. The process used for finding and confirming cases displayed by other sites may differ.
Surveillance Reports
Yes. CDC is modifying existing surveillance systems to track COVID-19. On April 3, we posted the first of what will be a weekly surveillance report called COVIDView. The report, updated each Friday, will summarize and interpret key indicators, including information related to COVID-19 outpatient visits, emergency department visits, hospitalizations and deaths, as well as laboratory test results.
In these weekly reports, CDC provides data on hospitalization rates and patient demographics as part of its COVID-NET surveillance system.
COVID-19 surveillance data are also used to produce publications, including CDC’s Morbidity and Mortality Weekly Report (MMWR), and to inform guidance documents to protect people from COVID-19 in a variety of settings.
CDC COVID Data Tracker
CDC COVID Data Tracker is a website that allows users to interact with a variety of data on COVID-19 that is updated daily. The website builds on other agency efforts – such as CDC’s new weekly COVID-19 surveillance report, COVIDView – to capture the impact the virus is having on the United States. CDC COVID Data Tracker presents data using visual dashboards that include interactive maps and graphs. It is being developed in phases. The first phase showcases data on U.S. COVID-19 cases and deaths as well as the social impacts of COVID-19.
CDC COVID Data Tracker is updated daily. Specifics of data reporting are described as a footnote on each page.
CDC COVID Data Tracker users can track the following data daily:
- U.S. cases and deaths of COVID-19 and the impacts of COVID-19.
- Trends in U.S. COVID-19 cases by jurisdiction, including case information over time and by age group and sex.
- Laboratory tests performed by U.S. commercial, hospital, public health, and CDC laboratories and the percentage of positive test results.
- School closures in the United States due to COVID-19, including information on planned dates of reopening, the percentage of school districts to remain closed over time, and projections for how long schools may remain closed (7 days, 14 days, 21 days, and 1 month)
- Impact of COVID-19 events, which includes news reports related to social impact events resulting from the COVID-19 outbreak in the United States, such as school closures, localized outbreaks, and state of emergency declarations.
- Mobility data from local areas including information on mobility data in the workplace, retail and recreation, transit station, and people staying at home.
CDC COVID Data Tracker displays these data using visual dashboards that include interactive maps and graphics. The data and information provided by COVID Data Tracker will be expanded over time.
- Syndromic surveillance showing trends in emergency department visits based on influenza-like-illness and COVID-19-like illness
- Global COVID-19 cases based on data provided by the World Health Organization (WHO)
- Global COVID-19 case trends based on WHO data